Atomic mass of carbon1/13/2024 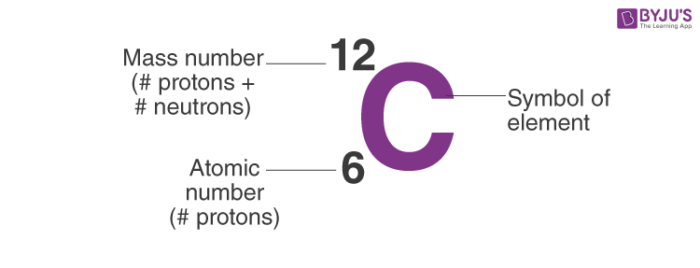
The unified atomic mass is defined as 1/ 12 of the mass of one atom of the nuclide 12C. The so-called unified atomic mass (unit symbol u) serves to quantify atomic and molecular mass. Clearly, weighing isn’t anymore an option. And even the attogram is roughly by a factor of 1000 larger than the mass of a single molecule of insulin. So let us go down by several orders of magnitude: 1 ng (nanogram, one billionth of a g or 10 –9 g), 1 pg (10 –12 g), 1 fg (femtogram, 10 –15 g), 1 ag (attogram, 10 –18 g). Even the most sensitive laboratory scales, capable of weighing down to about 1 µg (microgram, one millionth of a g or 10 –6 g) are far away from measuring the mass of a molecule. 
Now, the proportion of this grapefruit to Earth is also about 1:100.000.000. To illustrate the tiny size of a molecule, one may compare the proportions of a molecule of ascorbinic acid (vitamin C, ca. Large molecules, in particular macromolecules are composed of many atoms. Doing so yields 1.99 ´ 10 –26 kg as the mass of a carbon atom. To calculate the mass of a single atom of carbon, we just need to divide the molar mass of 12.0 g (0,012 kg) by the number of particles per mole (Avogadro’s number). The mole is defined as a number of particles, 6.022 ´ 10 23 particles to be accurate. The amount of 12.0 g of carbon corresponds to the amount of substance we define as one mole (1 mol). The masses of atoms and molecules are extremely small, by far beyond our imagination. However, their mass is still the same independent of where they are. We all know the examples of astronauts on Moon where they experience just 1/6 of their weight on Earth. Mass is a physical quantity that is independent of local gravitation. Strictly speaking, a pair of scales measures the weight of an object as the result of being subjected to the Earth’s gravitation. Commonly, we use scales for this type of measurement. Geological specimens are known in which the element has an isotopic composition outside the limits for normal material.We are perfectly used to the determination of the mass of a macroscopic object.See table 1 for details of range and original paper for the atomic weight of the element from different sources.However three such elements (Th, Pa, and U) do have a characteristic terrestrial isotopic composition, and for these an atomic weight is tabulated. , indicates the mass number of the longest-lived isotope of the element. Substantial deviations in atomic weight of the element from that given in the Table can occur. Modified isotopic compositions may be found in commercially available material because it has been subject to an undisclosed or inadvertant isotopic fractionation.Value being given the tabulated value should be applicable to any normal material. Range in isotopic composition of normal terrestrial material prevents a more precise.The difference between the atomic weight of the element in such specimens and that given in the Table may exceed the stated uncertainty. Geological specimens are known in which the element has an isotopic composition outside the limits for normal material.See original paper for the range of these elements from different sources List of Elements with Range of Atomic Weights. See also a copy of the periodic table with atomic weights to five significant figures. See below for the elements listed in Atomic Number Order or Name order. The original paper should be consulted for full details of the variation in atomic weight and the half life of the radioisotopes quoted below.Ī number in parentheses indicates the uncertainty in the last digit of the atomic weight. For radioactive elements the isotope with the longest half-life is quoted in parenthesis. In the other lists the values quoted are those suggested for material where the origin of the sample is unknown. The standard atomic weights of twelve elements having two or more stable isotopes have variability of atomic-weight values in natural terrestrial materials. Previous values may be consulted from the 1993 table, the 1995 table, the 1997 table, the 1999 table, the 2001 table, the 2005 table, the 2007 table, the 2009 table, the 2011 table, the 2013 table, the 2015 table or the 2019 table. World Wide Web version of atomic weight data originally prepared by G. These tables are based on the 2021 table with changes from the 2019 table for the values of Ar, Hf, Ir, Pb and Yb and changes to the uncertainty for Al, Au, Co, F, Ho, Mn, Nb, Pa, Pr, Rh, Sc, Tb, Tm, and Y. 2021 Atomic Weights IUPAC Commission on Isotopic Abundances and Atomic Weights.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
